 
The Bohr model was the first atomic model incorporating some quantum mechanics. Danish physicist Niels Bohr proposed the model in 1913. It’s called a planetary or cake model because electrons orbit the atomic nucleus like planets orbit the Sun, while the circular electron orbits form shells, like the layers of a cake. The Bohr model or Rutherford-Bohr model of the atom is a cake or planetary model that describes the structure of atoms mainly in terms of quantum theory. It is the first atomic model based mainly on quantum mechanics. The Bohr model is a cake or planetary model of the atom, with electrons in shells. He said the work has potential applications in next-generation computers and in the study of classical and quantum chaos.This entry was posted on Jby Anne Helmenstine (updated on May 10, 2021) "Our measurements show that the electrons remain localized for several orbits and behave much as classical particles," Dunning said. In fact, the atoms are true atomic giants, with diameters approaching one millimeter. Using a carefully tailored series of short electric pulses, the team was then able to coax the atoms into a precise configuration with one point-like, "localized" electron orbiting far from the nucleus. Using lasers, the researchers excited potassium atoms to extremely high levels. The team included members from Oak Ridge National Laboratory and Vienna University of Technology. 
"Using highly excited Rydberg atoms and a series of pulsed electric fields, we were able to manipulate the electron motion and create circular, planet-like states." "In a sufficiently large system, the quantum effects at the atomic scale can transition into the classical mechanics found in Bohr's model," said lead researcher Barry Dunning, Rice's Sam and Helen Worden Professor of Physics and Astronomy. 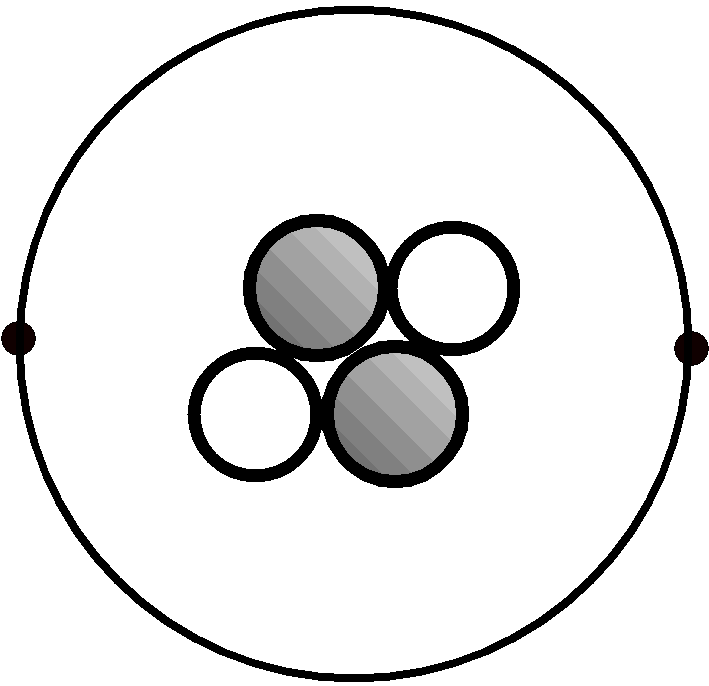
But his notion of electrons traveling in discrete orbits was eventually displaced by quantum mechanics, which revealed that electrons don't have precise positions but are instead distributed in wave-like patterns. Bohr's model led to a deeper understanding of both the chemical and optical properties of atoms and won him a Nobel Prize in 1922. The research is available online in Physical Review Letters.īohr offered the first successful theoretical model of the atom in 1913, suggesting that electrons traveled in orbits around the atom's nucleus like planets orbiting a star. HOUSTON - J- Nearly a century after Danish physicist Niels Bohr offered his planet-like model of the hydrogen atom, a Rice University-led team of physicists has created giant, millimeter-sized atoms that resemble it more closely than any other experimental realization yet achieved. Image: Using laser beams and electric fields, Rice physicists coaxed a point-like, "localized" electron to orbit far from the nucleus of a potassium atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
